Biofilm-protected bacterial infections are among the most persistent and costly challenges in modern medicine. Health Science is hard at work developing various solutions to the problem. This is my contribution. Estimates suggest that 65 to 80 percent of all clinical infections involve biofilm formation, and organisms embedded within these structures can tolerate antibiotic concentrations 100 to 1,000 times higher than their free-floating counterparts. The clinical consequences are familiar to anyone who has managed chronic wounds, implant-associated infections, or recurrent urinary tract infections: repeated courses of escalating antibiotics, diminishing returns, growing resistance, and limited options.
(Dropdown): Additional Context
Current strategies for addressing biofilms tend to rely on expensive, slow, and increasingly outpaced drug development pipelines. I offer the following. What makes this theoretical framework worth attention is not that it proposes a new compound, but that it identifies a potentially powerful combination hiding in plain sight. EGCG, the primary catechin in green tea, has well-documented antimicrobial and antibiotic-sensitizing properties. N-acetylcysteine (NAC), a widely available mucolytic and antioxidant, has demonstrated the capacity to physically dismantle biofilm architecture. Both are inexpensive, accessible, and carry established safety profiles.
They have never been studied together in an antimicrobial context, likely because a surface-level pharmacological analysis suggests they would cancel each other out. This paper argues that analysis is incomplete and that the combination may represent a multi-mechanism adjunct strategy capable of addressing biofilm shielding and enzymatic resistance simultaneously. If the mechanistic logic holds under experimental testing, it could offer clinicians a low-cost, readily available addition to the antimicrobial toolkit at a time when that toolkit is running dangerously thin.
A Theoretical Framework for EGCG-NAC Combinatorial Strategy Against Biofilm-Protected Bacterial Infections
-
Highlights
-
Abstract
-
Full Text
-
PDF / Download
-
Legal & Contact
Clinical Significance:
Biofilm-mediated bacterial infections account for 65 to 80 percent of clinical infections and exhibit 100- to 1,000-fold increased antibiotic tolerance, representing one of the most persistent challenges in infectious disease management. This theoretical framework identifies a combinatorial strategy using two well-characterized, accessible compounds (EGCG and NAC) that may address the two primary barriers to biofilm infection treatment simultaneously: physical shielding and enzymatic resistance.
Novelty:
As far as I can tell, this is the first framework proposing the deliberate combination of EGCG and NAC as a dual-adjunct antimicrobial strategy. Despite extensive independent literature on EGCG's antimicrobial properties and NAC's biofilm-disrupting capacity, this combination appears to have been overlooked due to a surface-level pharmacological concern (antioxidant quenching of pro-oxidant activity) that, on a multi-mechanism analysis, accounts for only one of at least four (probably more) interaction categories. The EGCG-2'-NAC conjugate, previously characterized only in oncology research, has never been examined in an antimicrobial context and represents the central testable hypothesis.
Translational Relevance:
Both compounds carry favorable regulatory profiles. NAC is FDA-approved, and EGCG holds Generally Recognized as Safe (GRAS) status. This positions the combination for a shorter translational pathway than novel drug development, with topical applications (chronic wounds, surgical sites, catheter-associated infections) representing the most immediate clinical targets.
Future Directions: This framework demonstrates the need to:
- Characterize the EGCG-2'-NAC conjugate in antimicrobial systems to determine whether it retains, loses, or amplifies antibacterial activity relative to either parent compound.
- Model conjugate formation kinetics under biofilm-relevant microenvironmental conditions (acidic pH, variable oxygen, thiol concentration gradients).
- Evaluate sequential versus simultaneous delivery against clinically relevant biofilm-forming organisms, including MRSA, carbapenem-resistant Enterobacteriaceae, and multidrug-resistant Pseudomonas aeruginosa.
- Investigate formulation strategies (nanoparticle encapsulation, timed-release systems) that optimize the phased delivery the model predicts is most effective.
Biofilm-mediated antibiotic resistance represents one of the most intractable challenges in clinical infectious disease management. Biofilms, the structured microbial communities encased in a self-produced extracellular polymeric substance (EPS) matrix, confer tolerance to antimicrobial agents that is orders of magnitude greater than that observed in planktonic bacteria. Two well-characterized compounds, epigallocatechin-3-gallate (EGCG) and N-acetylcysteine (NAC), have each demonstrated independent antimicrobial and antibiofilm properties through distinct mechanistic pathways. However, their potential as a combinatorial strategy has remained unexplored, likely due to a surface-level pharmacological concern: NAC, as a thiol antioxidant, would be expected to quench the pro-oxidant bactericidal activity of EGCG, effectively neutralizing one compound with the other. This paper argues that this concern, while mechanistically valid in isolation, reflects an incomplete analysis that examines only one of several independent antimicrobial mechanisms each compound employs. When the full mechanistic profiles of both compounds are layered, a complementary model emerges in which NAC degrades biofilm architecture by disrupting disulfide bonds, while EGCG disarms resistance enzymes and disrupts bacterial membranes through non-oxidative pathways. The oxidative "cancellation" represents, at most, the loss of a single kill mechanism in exchange for dramatically enhanced penetration, sensitization, and structural exposure of the target organisms. Furthermore, the documented formation of an EGCG-NAC thiol conjugate (EGCG-2’-NAC) with enhanced redox activity introduces the central testable hypothesis of this framework: that the interaction may, under specific microenvironmental conditions, amplify rather than diminish pro-oxidant bacterial killing. This paper presents a theoretical framework for a sequential, multi-phase assault model, examines species-specific and mixed-infection considerations, and proposes specific experimental and computational research directions to evaluate its clinical potential.
Introduction
The clinical management of biofilm-associated infections stands as one of the most consequential unsolved problems in contemporary medicine. Biofilms account for an estimated 65% to 80% of all bacterial infections treated in clinical settings, including chronic wound infections, prosthetic device infections, catheter-associated urinary tract infections, chronic prostatitis, chronic rhinosinusitis, and endocarditis (Costerton et al., 1999; Hall-Stoodley et al., 2004). The defining characteristic of biofilm-mediated infection is therapeutic recalcitrance: organisms embedded within biofilm architecture demonstrate antibiotic tolerance that is 100 to 1,000 times greater than their planktonic counterparts (Stewart & Costerton, 2001). This tolerance is not primarily genetic resistance but rather a phenotypic state conferred by the biofilm’s extracellular polymeric substance (EPS) matrix, which functions as a physical diffusion barrier, a chemical neutralization zone, and a metabolic sanctuary that reduces bacterial susceptibility to agents requiring active cellular processes for efficacy.
Current therapeutic approaches to biofilm-associated infections rely overwhelmingly on prolonged, high-dose antibiotic regimens that frequently fail and that contribute to the broader crisis of antimicrobial resistance. The development of adjunctive strategies capable of disrupting biofilm architecture while simultaneously enhancing antibiotic efficacy has become an urgent priority. Two compounds with extensive safety profiles and documented antibiofilm properties have emerged from independent research traditions: epigallocatechin-3-gallate (EGCG), the principal catechin of green tea, and N-acetylcysteine (NAC), a clinically approved mucolytic and antioxidant. Each has been studied as an individual antibiotic adjunct with promising results. Their combination, however, appears to have been overlooked entirely.
The most likely explanation for this gap is a reasonable but incomplete pharmacological inference. EGCG exerts a significant portion of its bactericidal activity through pro-oxidant mechanisms, specifically the generation of reactive oxygen species (ROS) and hydrogen peroxide that overwhelm bacterial antioxidant defenses (Arakawa et al., 2004). NAC, as a thiol antioxidant and glutathione precursor, would be expected to scavenge precisely those reactive species, neutralizing the oxidative killing mechanism. From this perspective, co-administration appears self-defeating: one agent cancels the other. This paper argues that this analysis, while pharmacologically accurate for one specific mechanism, is critically incomplete. It fails to account for the multiple independent antimicrobial and antibiofilm mechanisms each compound employs, the distinct molecular targets each addresses, and the possibility that their interaction produces a novel conjugate with enhanced, rather than diminished, biological activity. More pointedly, this paper identifies the EGCG-2’-NAC conjugate, characterized in oncology research by Lambert et al. (2008) but never examined in an antimicrobial context, as the central testable hypothesis that could resolve the apparent pharmacological paradox.
Background: Independent Mechanisms of Action
EGCG as an Antimicrobial Agent
EGCG demonstrates broad-spectrum antimicrobial activity through at least four mechanistically distinct pathways. First, EGCG binds directly to bacterial lipid membranes, causing structural damage, pore formation, and intramembranous leakage (Cui et al., 2012; Steinmann et al., 2013). This membrane disruption is substantially more pronounced against Gram-positive organisms, whose exposed peptidoglycan layer permits direct catechin-membrane interaction, with minimum inhibitory concentrations (MICs) against Gram-negative bacteria typically 8- to 16-fold higher than those for Gram-positive species (Reygaert, 2014). The Gram-negative outer membrane, composed primarily of negatively charged lipopolysaccharides, provides a partial shield against EGCG penetration. Second, EGCG inhibits specific reductases (FabG and FabI) in bacterial type II fatty acid synthesis, disrupting a biosynthetic pathway essential for membrane maintenance (Zhang & Rock, 2004). Third, EGCG inhibits dihydrofolate reductase, the same enzymatic target exploited by trimethoprim, thereby interfering with folate metabolism and nucleotide synthesis (Navarro-Martinez et al., 2005). Fourth, EGCG generates ROS through auto-oxidation, producing up to two molecules of hydrogen peroxide per EGCG molecule at physiological pH, which induces oxidative damage to bacterial macromolecules (Arakawa et al., 2004).
Beyond direct antimicrobial activity, EGCG demonstrates clinically significant synergy with conventional antibiotics, particularly against resistant organisms. Zhao et al. (2002) demonstrated that EGCG at 12.5 micrograms per milliliter in combination with penicillin produced synergistic effects against 100% of 21 MRSA strains tested. The mechanism involves dose-dependent inhibition of penicillinase production, effectively disarming the beta-lactamase resistance mechanism and restoring antibiotic sensitivity. Stapleton et al. (2004) confirmed that the gallate moiety is essential for this oxacillin-modulating activity. Notably, the development of lipid-soluble EGCG derivatives (EGCG-S) has addressed the stability limitations of native EGCG. Yussof et al. (2019) demonstrated that EGCG-S enhanced antibiotic efficacy by 18% to 112% across multiple antibiotic-organism combinations, converting resistant organisms to susceptible in numerous instances. EGCG-S combined with antibiotics also achieved 95% to 99% inhibition of biofilm formation across E. coli, Pseudomonas aeruginosa, S. aureus, S. epidermidis, and M. smegmatis (Shinde et al., 2021). The improved stability and bioavailability of lipid-soluble derivatives is directly relevant to the conjugate considerations discussed later in this paper, as chemical stabilization strategies that preserve EGCG’s reactive catechol groups may similarly influence conjugate formation kinetics and longevity.
NAC as a Biofilm Disruptor
NAC’s antibiofilm properties operate through mechanisms fundamentally different from its well-known antioxidant and mucolytic functions. The thiol (sulfhydryl) group on NAC reduces disulfide bonds in the structural proteins of the biofilm EPS matrix, directly dismantling the cross-linked protein scaffold that maintains biofilm architecture (Li et al., 2020; Dinicola et al., 2014). This is not an indirect or secondary effect; it is a direct chemical interaction between NAC’s reactive thiol and the cysteine-cysteine disulfide bridges that stabilize EPS structural proteins. Pseudomonas aeruginosa, one of the most clinically significant biofilm-forming organisms, relies heavily on disulfide-bonded proteins in its EPS, including the amyloid protein FapC (which requires two conserved cysteines for fibril formation), extracellular lipases, and elastases (Li et al., 2020). This pronounced efficacy against Pseudomonas-dominated biofilms is a consistent finding across multiple research groups and clinical contexts.
NAC’s antibiofilm activity extends beyond disulfide bond disruption. It reduces extracellular polysaccharide production, cleaves extracellular DNA (eDNA) that forms part of the biofilm matrix, and creates local pH changes that further destabilize the EPS (Jotic et al., 2025). In chronic wound biofilm models using bacteria harvested directly from infected wounds rather than laboratory-cultured strains, NAC at 20 mg/mL dismantled mature Pseudomonas-dominated biofilms within 24 hours (Li et al., 2020). A systematic review of clinical trials concluded that NAC in combination with antibiotics significantly promoted antibiotic permeability to the deepest layers of biofilms, overcoming biofilm-mediated resistance (Dinicola et al., 2014). This adjunctive capacity, enhancing antibiotic penetration by degrading the structural barrier, distinguishes NAC from agents that merely inhibit biofilm formation without addressing established biofilm architecture.
NAC’s biofilm-disrupting properties have been demonstrated across a broad range of organisms and clinical contexts, including Staphylococcal biofilms in chronic rhinosinusitis (Jotic et al., 2025), Pseudomonas aeruginosa biofilms in chronic wounds and cystic fibrosis airways (Li et al., 2020), Candida biofilms in fungal infections, and mixed-species biofilms on implanted medical devices. Its mechanism of action is largely independent of bacterial strain or species because it targets the conserved structural chemistry of the EPS matrix rather than organism-specific metabolic pathways. However, efficacy is dose- and matrix-composition-dependent; Staphylococcal biofilms that rely more heavily on polysaccharide intercellular adhesin (PIA) than on disulfide-bonded proteins may require higher NAC concentrations for equivalent disruption (Jotic et al., 2025).
The EGCG-NAC Conjugate: The Central Hypothesis
Lambert et al. (2008) identified a previously uncharacterized chemical entity formed when EGCG and NAC interact under biological conditions. When EGCG undergoes oxidation to its quinone form, the activated carbon can react with thiol-containing compounds. In the presence of NAC, this produces EGCG-2’-NAC, a thiol conjugate with biological properties distinct from either parent compound. In lung cancer cell models, this conjugate demonstrated enhanced rather than diminished cytotoxic activity. NAC dose-dependently enhanced the growth inhibitory activity of EGCG, producing an 8.8-fold increase in apoptosis compared to either agent alone. NAC also increased intracellular EGCG levels 5.5-fold and extended EGCG’s half-life from 8.5 hours to 22.7 hours. Critically, Lambert et al. (2008) observed that incubation of the related EGCG-cysteine conjugates under cell culture conditions resulted in hydrogen peroxide generation at a rate faster than equimolar concentrations of EGCG alone, suggesting the conjugate family is more redox-active than the parent compound.
This finding has not, to date, been examined in an antimicrobial context. The conjugate was characterized exclusively in oncology research, and its potential relevance to infectious disease has not been explored. This represents the most significant gap identified in this paper, and its resolution constitutes the central testable hypothesis of the proposed framework. The primary bactericidal mechanism of EGCG against both Gram-positive and Gram-negative organisms involves intracellular ROS generation and oxidative stress (Xiong et al., 2017; Arakawa et al., 2004). A conjugate that generates ROS at an accelerated rate, as the EGCG-thiol conjugates appear to do, could in principle amplify rather than diminish this killing mechanism. Whether this occurs in practice depends on variables that have not been measured: the rate and location of conjugate formation in the presence of bacterial cells and biofilm matrices, the conjugate’s membrane permeability and intracellular accumulation in bacterial versus mammalian cells, and its stability under the pH conditions characteristic of biofilm microenvironments (which are typically more acidic than surrounding tissue). These variables are empirically tractable and should be prioritized in any experimental program designed to evaluate this framework.
The lipid-soluble EGCG derivative (EGCG-S) characterized by Yussof et al. (2019); Shinde et al. (2021) offers a relevant precedent. Native EGCG is water-soluble and relatively unstable, with rapid degradation limiting its bioavailability and effective concentration at target sites. EGCG-S, produced by esterification with stearic acid, demonstrated substantially improved stability while retaining antimicrobial and antibiofilm properties. An analogous approach, designing thiol conjugate derivatives or stabilized EGCG formulations that favor controlled conjugate formation at the target site, could address the stability and timing challenges inherent in the proposed model.
The Apparent Paradox and Its Resolution
The Single-Lens Problem
The assumption that NAC would neutralize EGCG’s antimicrobial activity rests on a single mechanistic observation: EGCG kills bacteria partly through pro-oxidant ROS generation, and NAC scavenges ROS. This has been directly confirmed. When researchers added NAC to EGCG-treated E. coli cultures, the bactericidal effect was reduced, because NAC quenched the reactive oxygen species that EGCG was generating intracellularly (Xiong et al., 2017). Under anaerobic conditions, EGCG’s inhibitory effect was similarly prevented, confirming the oxygen-dependent nature of this specific killing pathway. Taken in isolation, these results would reasonably discourage further investigation of the combination.
This reasoning, however, commits what might be termed a single-lens analytical error. It evaluates the interaction through only one of the several independent antimicrobial mechanisms each compound employs, then generalizes from that single interaction to the entire pharmacological relationship. The error is analogous to concluding that a military force is ineffective because one of its five weapons systems was disabled, without assessing the status or capability of the remaining four. A more complete analysis requires mapping the full mechanism portfolio of each compound and evaluating the interaction at each mechanistic level independently.
Multi-Mechanism Interaction Analysis
When the complete mechanism profiles of EGCG and NAC are layered, the interaction landscape is substantially more complex than the single-lens analysis suggests. Four categories of interaction emerge.
Category 1: Antagonistic interaction (oxidative killing). NAC does quench EGCG’s extracellular ROS generation. This reduces one of EGCG’s four bactericidal mechanisms. This is the only interaction the single-lens analysis captures, and it is real. However, it represents the loss of one mechanism among many, not the neutralization of the entire antimicrobial program.
Category 2: Independent, parallel activity (membrane disruption, enzyme inhibition). EGCG’s direct binding to bacterial lipid membranes, its inhibition of fatty acid synthesis enzymes, and its inhibition of dihydrofolate reductase are not mediated by ROS and are therefore unaffected by NAC’s antioxidant activity. These mechanisms operate through direct molecular binding interactions, not oxidative chemistry. They continue to function in full whether NAC is present or absent.
Category 3: Complementary, synergistic activity (biofilm disruption plus resistance disarmament). This is the most consequential category for established infections. NAC degrades the biofilm EPS matrix through disulfide bond reduction, exposing organisms that were previously shielded. EGCG inhibits penicillinase and other resistance enzymes, restoring antibiotic sensitivity in organisms that were previously resistant. These two effects are not merely additive; they are sequentially synergistic. NAC removes the physical barrier (biofilm), and EGCG removes the enzymatic barrier (resistance mechanisms), creating a two-stage vulnerability in organisms that previously possessed both forms of protection.
Category 4: Potentially amplifying interaction (conjugate formation). The EGCG-2’-NAC conjugate identified by Lambert et al. (2008) is more redox-active than EGCG alone. If this conjugate forms at or near the bacterial surface after biofilm disruption has begun to expose organisms, the pro-oxidant killing mechanism may not only be preserved but enhanced. The timing and location of conjugate formation become critical variables. Extracellular conjugation in free solution would produce antioxidant quenching (Category 1). Conjugation at the bacterial membrane or intracellularly, after EGCG has already engaged its target, could produce amplified oxidative killing (Category 4). These are not mutually exclusive outcomes; both may occur simultaneously, with the net effect determined by the relative kinetics of each reaction. Characterizing these kinetics, ideally through both in vitro assay and computational modeling, represents the highest-priority research question generated by this framework.

Figure 1. Four categories of EGCG-NAC mechanistic interaction. The single-lens analysis captures only Category 1 (antagonistic), overlooking three additional interaction categories that preserve or enhance antimicrobial efficacy.
Proposed Model: Sequential Multi-Phase Assault
Based on the multi-mechanism interaction analysis, this paper proposes a three-phase sequential assault model for the EGCG-NAC combination as an adjunct to conventional antibiotic therapy against biofilm-protected infections.
Phase 1: Structural Degradation (NAC-Driven)
NAC initiates the assault through direct chemical attack on the biofilm EPS matrix. The thiol group reduces disulfide bonds in structural proteins, dismantling the cross-linked scaffold. Extracellular polysaccharide production is reduced. Extracellular DNA is cleaved. The local pH shift further destabilizes the matrix. The net effect is progressive thinning and fragmentation of the biofilm architecture, increasingly exposing the bacterial organisms embedded within. This phase exploits NAC’s established biofilm-disrupting capacity and is entirely independent of any interaction with EGCG. It operates whether EGCG is present or absent, and its efficacy is unaffected by the oxidative interaction between the two compounds.
Phase 2: Resistance Disarmament and Membrane Disruption (EGCG-Driven)
As the biofilm matrix degrades and organisms become increasingly exposed, EGCG engages through its non-oxidative mechanisms. Direct binding to bacterial lipid membranes causes structural damage, pore formation, and leakage. Inhibition of penicillinase disarms beta-lactam resistance, restoring sensitivity to penicillins, cephalosporins, and carbapenems. Inhibition of fatty acid synthesis and dihydrofolate reductase further compromises bacterial cellular maintenance. These mechanisms do not require ROS generation and are therefore fully operational regardless of NAC’s antioxidant activity. The organisms are now structurally exposed (Phase 1), membrane-compromised, and resistance-disarmed (Phase 2).
Phase 3: Lethal Delivery (Conventional Antibiotic, with Potential Conjugate Amplification)
The conventional antibiotic, now encountering organisms stripped of both their physical (biofilm) and enzymatic (resistance mechanism) defenses, can engage its primary target at effective concentrations for the first time. Beta-lactams reach penicillin-binding proteins without penicillinase degradation. Fluoroquinolones access DNA gyrase without the diffusion barrier of intact EPS. Aminoglycosides penetrate membranes already compromised by EGCG. If the EGCG-NAC conjugate forms at the bacterial surface during this phase, its enhanced redox activity adds an additional oxidative assault against organisms that have already lost their structural protection and whose membranes have already been compromised, maximizing the impact of ROS on vulnerable targets.
The critical insight of this model is that even under the worst-case assumption, where NAC completely eliminates EGCG’s pro-oxidant killing and no conjugate-mediated amplification occurs, the remaining mechanisms still constitute a dual-adjunct platform that simultaneously addresses the two primary barriers to treating biofilm-associated infections: the physical barrier of the biofilm itself and the enzymatic barrier of antibiotic resistance. The oxidative "loss" is exchanged for dramatically enhanced delivery and sensitization. In the context of chronic, biofilm-protected infections where oxidative stress against shielded organisms is largely wasted effort anyway (the EPS absorbs and neutralizes much of it before it reaches the bacteria), this trade is not merely acceptable; it is strategically advantageous.
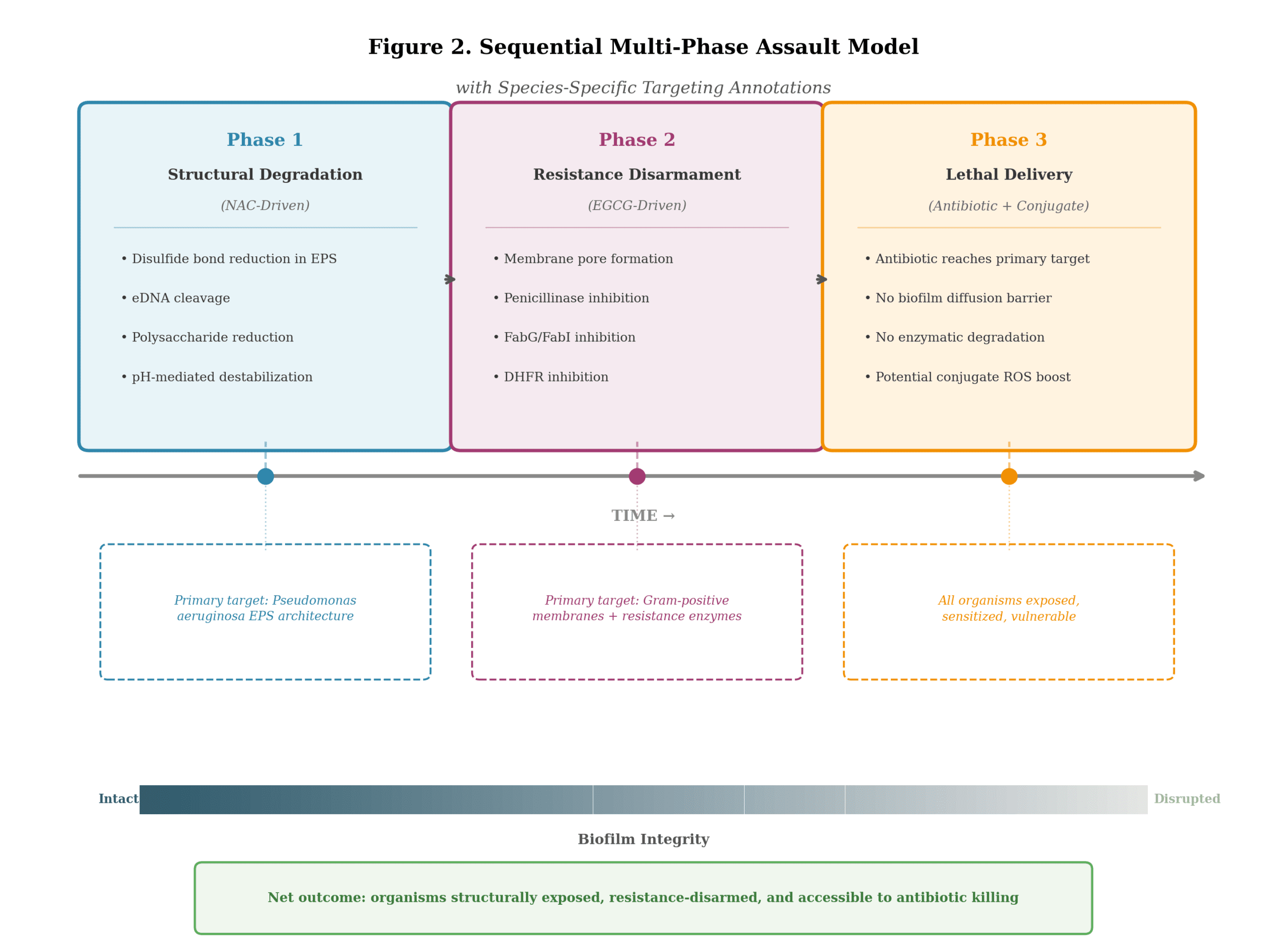
Figure 2. Sequential multi-phase assault model showing NAC-driven structural degradation (Phase 1), EGCG-driven resistance disarmament (Phase 2), and antibiotic lethal delivery with potential conjugate amplification (Phase 3), annotated with species-specific targeting preferences.
Species-Specific and Mixed-Infection Considerations
A critical consideration for clinical translation is the differential susceptibility of Gram-positive and Gram-negative organisms to each compound, and the implications for the mixed-species biofilms that characterize most clinical infections. EGCG and NAC do not exert equivalent effects across all organisms, and the combinatorial model must account for this heterogeneity.
EGCG’s direct antimicrobial effects are substantially stronger against Gram-positive bacteria. Minimum inhibitory concentrations against Gram-negative organisms are typically 8- to 16-fold higher than those for Gram-positive species (Reygaert, 2014), because the Gram-negative outer membrane and its lipopolysaccharide component impede EGCG access to the cytoplasmic membrane and intracellular targets. Conversely, EGCG’s antibiotic-synergy effects, particularly penicillinase inhibition, are most consequential against Gram-positive organisms like MRSA where beta-lactam resistance is clinically prevalent. NAC’s biofilm disruption, by contrast, has demonstrated its most robust effects against Pseudomonas aeruginosa, a Gram-negative organism whose EPS is particularly dependent on disulfide-bonded structural proteins (Li et al., 2020). Staphylococcal biofilms, which may rely more heavily on PIA-based (polysaccharide intercellular adhesin) matrices than on disulfide-bonded proteins, show more variable susceptibility to NAC and may require higher concentrations for equivalent disruption (Jotic et al., 2025). An open question with direct experimental implications is whether the EGCG-2’-NAC conjugate alters this Gram-positive/Gram-negative MIC asymmetry. If the conjugate’s enhanced redox activity translates to greater membrane-disruptive capacity, it could partially compensate for the outer membrane barrier that limits native EGCG’s efficacy against Gram-negative organisms, narrowing the 8- to 16-fold MIC gap. Conversely, if the conjugate’s larger molecular size impedes outer membrane penetration, the asymmetry could widen. This variable should be explicitly measured in the Priority 1 conjugate characterization studies proposed below.
This differential susceptibility profile, rather than being a limitation, may represent a strategic advantage in mixed-species biofilm infections. Chronic wound biofilms, prosthetic joint infections, and catheter-associated infections commonly involve polymicrobial communities containing both Gram-positive (S. aureus, S. epidermidis, Enterococcus species) and Gram-negative (P. aeruginosa, E. coli, Klebsiella species) organisms embedded within a shared or hybrid EPS matrix. In such mixed-species contexts, NAC’s strongest structural attack preferentially degrades the Pseudomonas-contributed EPS scaffolding while EGCG’s strongest direct killing and resistance disarmament preferentially targets the Gram-positive members. The net effect is a two-pronged strategy where each compound addresses the organisms most amenable to its mechanism of action, collectively covering a broader taxonomic range than either agent alone.
This complementary targeting has a second-order benefit. In polymicrobial biofilms, disruption of one species’ contribution to the shared EPS matrix often destabilizes the entire community structure. Pseudomonas aeruginosa frequently serves as a primary architectural organism in mixed biofilms, producing the alginate and structural proteins that other community members exploit for protection. NAC-mediated degradation of Pseudomonas-contributed EPS may therefore expose Gram-positive organisms that were sheltering within the Pseudomonas-built matrix, rendering them newly accessible to EGCG’s Gram-positive-preferential mechanisms. The sequential model thus predicts a cascading vulnerability in mixed-species contexts: NAC dismantles the structural framework that the Gram-negative organisms contributed, and EGCG kills the Gram-positive organisms that this framework was protecting.
Implications for Resistance Development
A significant theoretical advantage of the proposed combinatorial model is its potential impact on resistance evolution. Bacteria develop resistance to single-mechanism antibiotics because evolutionary pressure requires adaptation at only one molecular target. A beta-lactamase gene, a porin mutation, an efflux pump upregulation: each represents a single genetic event that confers survival advantage against a single-mechanism agent. The selection pressure is focused, and the evolutionary solution is correspondingly tractable.
The EGCG-NAC-antibiotic combination applies selection pressure across multiple independent targets simultaneously. To survive the proposed three-phase assault, an organism would need to: (a) modify its EPS structural proteins to resist thiol-mediated disulfide bond reduction by NAC; (b) alter its membrane lipid composition to resist EGCG binding and pore formation; (c) develop alternative resistance mechanisms to replace the penicillinase activity inhibited by EGCG; (d) maintain or restore fatty acid synthesis and folate metabolism pathways despite EGCG interference; and (e) retain conventional resistance to the antibiotic agent itself. The probability of simultaneous mutations addressing all five targets in a single lineage is exponentially lower than the probability of any single resistance mutation arising. This is the same evolutionary logic that underlies combination antiretroviral therapy for HIV, where simultaneous pressure on multiple independent viral targets renders escape mutations statistically improbable.
Furthermore, NAC’s mechanism of action targets the conserved chemistry of disulfide bonds in structural proteins, not an organism-specific metabolic pathway. Bacteria cannot easily evolve away from disulfide-bonded structural proteins because the disulfide bond is a fundamental biochemical mechanism for protein stabilization and function. Resistance to NAC’s biofilm-disrupting activity would require wholesale reorganization of EPS protein architecture, a far more genetically costly adaptation than a single point mutation in a drug target.
Proposed Research Directions
The theoretical framework presented here generates several testable hypotheses. They are presented in order of priority, with the conjugate characterization identified as the foundational experiment upon which the remaining investigations depend.
Priority 1: Characterization of the EGCG-NAC Conjugate in Antimicrobial Systems
The EGCG-2’-NAC conjugate has been characterized only in the context of cancer cell cytotoxicity. Its antimicrobial activity, redox behavior in the presence of bacterial cells, membrane interaction properties, and minimum inhibitory concentrations against clinically significant organisms (including MRSA, P. aeruginosa, and Enterococcus species) have not been determined. This represents the most fundamental gap in the current evidence base and the single experiment most likely to resolve the framework’s central uncertainty. Specific assays should include: (a) synthesis and isolation of the EGCG-2’-NAC conjugate in sufficient quantity for antimicrobial testing, following the methods described by Lambert et al. (2008); (b) MIC and minimum bactericidal concentration (MBC) determination against Gram-positive and Gram-negative panels; (c) measurement of intracellular ROS generation in bacterial cells exposed to the conjugate versus equimolar EGCG, using fluorescent ROS indicators; and (d) time-kill kinetics comparing the conjugate to EGCG alone and to the EGCG-NAC mixture.
If the conjugate demonstrates equal or greater antimicrobial activity compared to EGCG alone, the Category 4 (amplifying) interaction is validated and the entire framework shifts from speculative to empirically grounded. If the conjugate is antimicrobially inactive, the framework still holds through Categories 2 and 3, but the model simplifies to a dual-mechanism adjunct platform without the oxidative amplification component.
Priority 2: Conjugate Formation Kinetics and Microenvironmental Determinants
The critical variable determining whether the EGCG-NAC interaction produces net antioxidant quenching or net pro-oxidant amplification is the location and timing of conjugate formation relative to the bacterial target. Studies examining conjugate formation rates at different pH values (particularly the acidic pH ranges of 5.5 to 6.5 characteristic of biofilm interiors), in the presence of bacterial membrane components versus in free solution, and under conditions that mimic the thiol-rich microenvironment within a partially degraded biofilm would clarify whether the amplifying interaction is achievable in practice.
Computational modeling should accompany these empirical studies. In silico kinetic models simulating the competing reactions (NAC scavenging of EGCG-generated ROS versus NAC-EGCG conjugate formation versus EGCG auto-oxidation) at varying concentration ratios, pH values, and spatial distributions could predict the conditions under which amplification dominates over quenching. Reaction-diffusion models of biofilm penetration, parameterized with empirical diffusion coefficients for NAC and EGCG through biofilm matrices of varying density and composition, could further predict the spatial and temporal profile of each compound’s activity within the biofilm structure. Such models would substantially increase the predictive power of the framework and guide the design of subsequent in vitro experiments.
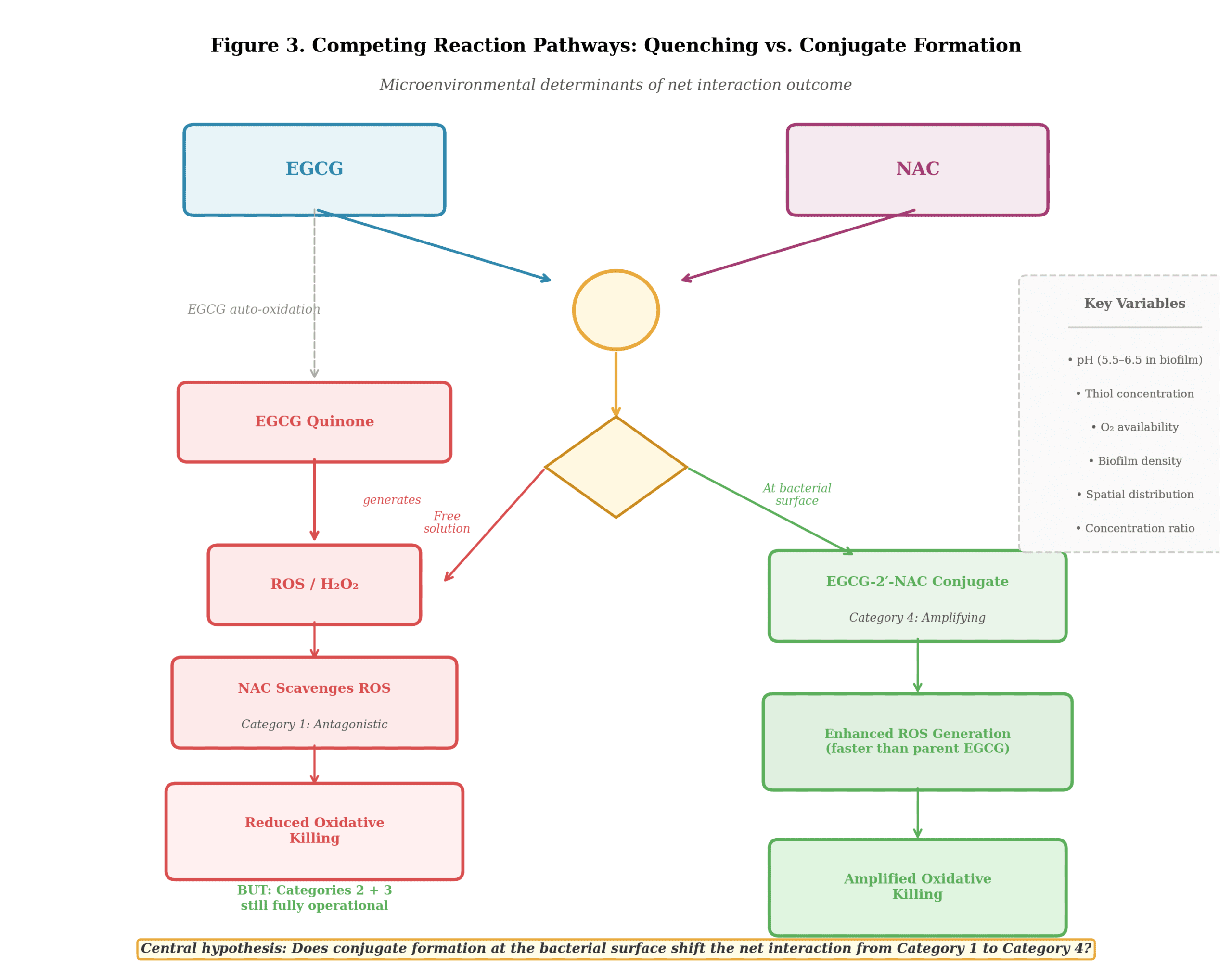
Figure 3. Competing reaction pathways illustrating how microenvironmental variables (pH, thiol concentration, spatial distribution) determine whether EGCG-NAC interaction produces net quenching (Category 1) or net amplification (Category 4) at the bacterial target.
Priority 3: Biofilm Penetration and Sequential Disruption Dynamics
Time-course studies examining the sequential effects of NAC-mediated EPS degradation followed by EGCG-mediated membrane disruption and resistance enzyme inhibition are needed to characterize the temporal dynamics of the proposed three-phase model. Confocal microscopy with fluorescent-tagged compounds could visualize the penetration sequence in real-time within established biofilm structures. Pre-treatment with NAC followed by EGCG exposure, versus simultaneous application, would test whether sequential delivery produces superior outcomes to co-administration, as the model predicts.
Priority 4: Formulation Strategies for Sequential Delivery
If conjugate formation location proves critical, formulation approaches that delay EGCG-NAC interaction until bacterial engagement has occurred could optimize the balance between biofilm disruption and oxidative killing. Encapsulation in lipid nanoparticles, timed-release coatings, or layered topical applications represent potential strategies. The work of Pinto et al. (2021) on NAC-loaded lipid nanoparticles functionalized with D-amino acids that targeted biofilm matrices provides a directly relevant model. Similar nanocarrier approaches could be adapted to deliver EGCG and NAC in a spatially or temporally controlled sequence. The improved stability observed with EGCG-S (Yussof et al., 2019) suggests that lipid modification of EGCG may also serve to modulate conjugate formation kinetics by altering the accessibility of the catechol ring to thiol attack.
Priority 5: Triple-Combination Efficacy and Resistance Development
The proposed model predicts that the EGCG-NAC combination should enhance the efficacy of conventional antibiotics against biofilm-forming, multidrug-resistant clinical isolates to a greater degree than either adjunct alone. Checkerboard synergy assays and time-kill studies using MRSA, carbapenem-resistant Enterobacteriaceae, and multidrug-resistant Pseudomonas aeruginosa biofilms would provide direct tests of this prediction. Mixed-species biofilm models, particularly those combining S. aureus with P. aeruginosa, would test the species-specific complementary targeting described above.
Serial passage resistance development experiments comparing resistance emergence rates in bacteria exposed to antibiotic alone versus antibiotic-plus-EGCG-NAC over multiple generations would test the theoretical prediction that multi-target pressure retards resistance evolution. These experiments should run for sufficient generations (typically 20 to 40 passages) to capture intermediate resistance phenotypes and should include whole-genome sequencing of resistant isolates to characterize the genetic basis of any resistance that does emerge.
Clinical Translation Considerations
Several features of the proposed combination make it unusually amenable to clinical translation relative to novel synthetic antimicrobial agents. Both NAC and EGCG possess established safety profiles with decades of human exposure data. NAC is an FDA-approved drug with well-characterized pharmacokinetics, dosing ranges, and adverse effect profiles. EGCG holds Generally Recognized as Safe (GRAS) status and has been consumed in green tea by billions of people across centuries. The regulatory pathway for a combination adjunct using two well-characterized compounds is substantially shorter and less costly than de novo drug development.
Topical applications represent the most immediately translatable formulation strategy. EGCG’s systemic bioavailability is limited, and the concentrations required for antimicrobial efficacy often exceed achievable plasma levels through oral administration (Steinmann et al., 2013). However, topical application to chronic wounds, surgical site infections, catheter surfaces, and implant-associated infections could readily achieve local concentrations within the effective range. NAC is already used topically in wound care and via nebulization for respiratory infections. A topical formulation combining both agents with a conventional antibiotic for application to biofilm-infected chronic wounds represents a near-term clinical development pathway.
Systemic applications, while more pharmacokinetically challenging, should not be dismissed. Oral NAC achieves meaningful systemic levels and is routinely used at therapeutic doses for its glutathione-replenishing and mucolytic properties. Oral EGCG supplements achieve detectable plasma concentrations, though at levels below the minimum inhibitory concentrations reported for most organisms. However, the proposed model does not require either compound to achieve bactericidal concentrations independently. The model relies on their adjunctive effects (biofilm disruption, resistance enzyme inhibition, membrane perturbation) at sub-MIC concentrations that are achievable systemically. The antibiotic, not the adjuncts, delivers the primary bactericidal effect; the adjuncts create the conditions under which the antibiotic can be effective.
Limitations and Counterarguments
This paper presents a theoretical framework supported by mechanistic reasoning and extrapolation from independent lines of evidence. It is not supported by direct experimental data testing the proposed combination in an antimicrobial context. The most significant limitation is the absence of empirical data on the EGCG-NAC conjugate’s antimicrobial properties. The conjugate has been characterized exclusively in oncology research, and its behavior in the context of bacterial cell membranes, biofilm microenvironments, and bacterial antioxidant systems may differ substantially from its behavior against mammalian cancer cells.
The in vitro evidence for NAC quenching EGCG’s oxidative bactericidal activity (Xiong et al., 2017) is direct and well-demonstrated. The argument that this loss is compensated by gains in other mechanistic categories is logical but untested. It is possible that the oxidative mechanism is more consequential for bactericidal activity than the remaining mechanisms, particularly for specific organisms or under specific growth conditions. The relative contribution of each individual mechanism to EGCG’s overall antimicrobial efficacy has not been fully partitioned, and the assumption that non-oxidative mechanisms provide adequate compensatory killing requires experimental validation.
Additionally, the pharmacokinetics of co-administered EGCG and NAC in vivo, where each compound would be absorbed at different rates, metabolized through different hepatic pathways, and distributed to target tissues at different concentrations and time points, may produce an interaction profile substantially different from what is predicted by in vitro combination studies. The sequential timing proposed in the three-phase model may be difficult to achieve in practice without sophisticated formulation strategies that control the temporal sequence of compound delivery to the infection site. Simultaneous exposure, the likely default in unformulated applications, may favor Category 1 (quenching) over Category 4 (amplification), potentially limiting the model’s practical utility unless formulation challenges are addressed.
Finally, the species-specific considerations identified in this paper, while framed as a potential advantage, also introduce complexity. The optimal EGCG:NAC ratio, the required antibiotic partner, and the expected efficacy may vary substantially depending on the organism(s) involved, the biofilm matrix composition, and the anatomical site of infection. A single fixed-dose combination is unlikely to be optimal across all clinical contexts, and organism-specific or site-specific formulation guidance would be needed for clinical implementation.
Conclusion
The EGCG-NAC combination as an antimicrobial biofilm strategy appears to have fallen into a research blind spot created by the intersection of two separate bodies of literature that have not communicated with each other. The antimicrobial EGCG researchers and the NAC biofilm researchers have each produced substantial evidence for the individual utility of their respective compounds, but the obvious pharmacological concern, that a thiol antioxidant would neutralize a pro-oxidant antimicrobial, has apparently prevented anyone from examining the combination through a wider lens.
When that wider lens is applied, the picture changes substantially. The oxidative cancellation is real but represents only one interaction among several. The remaining interactions are either neutral (preserving EGCG’s non-oxidative kill mechanisms), complementary (combining NAC’s structural attack on biofilm architecture with EGCG’s enzymatic disarmament of resistance mechanisms), or potentially amplifying (through a novel conjugate with enhanced redox activity). The trade, even under worst-case assumptions, exchanges a partially effective oxidative mechanism for dramatically improved target access and sensitization. In the clinical context of chronic biofilm-associated infections, where the biofilm itself is the primary obstacle to treatment, this is a favorable exchange. The addition of species-specific complementary targeting in mixed-infection contexts further strengthens the rationale for this combination.
The framework presented here does not claim to have identified a proven therapeutic strategy. It claims to have identified a gap in the literature created by disciplinary siloing and a surface-level pharmacological concern that, on deeper analysis, does not justify the neglect. The EGCG-2’-NAC conjugate, sitting unexamined at the intersection of oncology and infectious disease research, represents the most consequential unknown. Its characterization in antimicrobial systems should be the first experiment any team pursuing this direction conducts, as its outcome determines whether the model operates as a dual-mechanism adjunct (already a meaningful advance) or as a triple-mechanism platform with amplified oxidative killing. The proposed research directions are tractable, the compounds are accessible, and the clinical need is urgent. What remains is for someone to run the experiments. The EGCG-2’-NAC conjugate may prove to be the bridge between oncology and infectious disease that neither field knew it was missing.
References
Arakawa, H., Maeda, M., Okubo, S., & Shimamura, T. (2004). Role of hydrogen peroxide in bactericidal action of catechin. Biological and Pharmaceutical Bulletin, 27(3), 277–281. https://doi.org/10.1248/bpb.27.277
Costerton, J. W., Stewart, P. S., & Greenberg, E. P. (1999). Bacterial biofilms: A common cause of persistent infections. Science, 284(5418), 1318–1322. https://doi.org/10.1126/science.284.5418.1318
Cui, Y., Oh, Y. J., Lim, J., Youn, M., Lee, I., Pak, H. K., Park, W., Jo, W., & Park, S. (2012). AFM study of the differential inhibitory effects of the green tea polyphenol (−)-epigallocatechin-3-gallate (EGCG) against Gram-positive and Gram-negative bacteria. Food Microbiology, 29(1), 80–87. https://doi.org/10.1016/j.fm.2011.08.019
Dinicola, S., De Grazia, S., Carlomagno, G., & Pintucci, J. P. (2014). N-acetylcysteine as powerful molecule to destroy bacterial biofilms. A systematic review. European Review for Medical and Pharmacological Sciences, 18(19), 2942–2948.
Hall-Stoodley, L., Costerton, J. W., & Stoodley, P. (2004). Bacterial biofilms: From the natural environment to infectious diseases. Nature Reviews Microbiology, 2(2), 95–108. https://doi.org/10.1038/nrmicro821
Jotic, A., Cirkovic, I., Bozic, D., Savic Vujovic, K., Milovanovic, J., Folic, M., Trivic, A., Cvorovic, L., & Radivojevic, N. (2025). Antibiofilm effects of N-acetyl cysteine on Staphylococcal biofilm in patients with chronic rhinosinusitis. Microorganisms, 13(9), 2050. https://doi.org/10.3390/microorganisms13092050
Lambert, J. D., Sang, S., Hong, J., Kwon, S. J., Lee, M. J., Ho, C. T., & Yang, C. S. (2008). N-Acetylcysteine enhances the lung cancer inhibitory effect of epigallocatechin-3-gallate and forms a new adduct. Free Radical Biology and Medicine, 44(6), 1069–1074. https://doi.org/10.1016/j.freeradbiomed.2007.12.016
Li, X., Kim, J., Wu, J., Ahamed, A. I., Wang, Y., & Martins-Green, M. (2020). N-Acetyl-cysteine and mechanisms involved in resolution of chronic wound biofilm. Journal of Diabetes Research, 2020, 9589507. https://doi.org/10.1155/2020/9589507
Navarro-Martinez, M. D., Navarro-Peran, E., Cabezas-Herrera, J., Ruiz-Gomez, J., Garcia-Canovas, F., & Rodriguez-Lopez, J. N. (2005). Antifolate activity of epigallocatechin gallate against Stenotrophomonas maltophilia. Antimicrobial Agents and Chemotherapy, 49(7), 2914–2920. https://doi.org/10.1128/AAC.49.7.2914-2920.2005
Pinto, R. M., Lopes-de-Campos, D., Martins, M. C. L., Van Dijck, P., Nunes, C., & Reis, S. (2021). N-Acetyl-l-cysteine-loaded nanosystems as a promising therapeutic approach toward the eradication of Pseudomonas aeruginosa biofilms. ACS Applied Materials and Interfaces, 13(36), 42329–42340. https://doi.org/10.1021/acsami.1c05124
Reygaert, W. C. (2014). The antimicrobial possibilities of green tea. Frontiers in Microbiology, 5, 434. https://doi.org/10.3389/fmicb.2014.00434
Shinde, S., Lee, L. H., & Chu, T. (2021). Inhibition of biofilm formation by the synergistic action of EGCG-S and antibiotics. Antibiotics, 10(2), 102. https://doi.org/10.3390/antibiotics10020102
Stapleton, P. D., Shah, S., Anderson, J. C., Hara, Y., Hamilton-Miller, J. M., & Taylor, P. W. (2004). Modulation of beta-lactam resistance in Staphylococcus aureus by catechins and gallates. International Journal of Antimicrobial Agents, 23(5), 462–467. https://doi.org/10.1016/j.ijantimicag.2003.09.027
Steinmann, J., Buer, J., Pietschmann, T., & Steinmann, E. (2013). Anti-infective properties of epigallocatechin-3-gallate (EGCG), a component of green tea. British Journal of Pharmacology, 168(5), 1059–1073. https://doi.org/10.1111/bph.12009
Stewart, P. S., & Costerton, J. W. (2001). Antibiotic resistance of bacteria in biofilms. The Lancet, 358(9276), 135–138. https://doi.org/10.1016/S0140-6736(01)05321-1
Xiong, L. G., Chen, Y. J., Tong, J. W., Huang, J. A., Li, J., Gong, Y. S., & Liu, Z. H. (2017). Tea polyphenol epigallocatechin gallate inhibits Escherichia coli by increasing endogenous oxidative stress. Food Chemistry, 217, 196–204. https://doi.org/10.1016/j.foodchem.2016.08.098
Yussof, A., Habiba, U., Liaw, D., Chu, T., & Lee, L. H. (2019). Epigallocatechin gallate-stearate enhances the efficacy of antibiotics. Open Journal of Medical Microbiology, 9(3), 77–94. https://doi.org/10.4236/ojmm.2019.93009
Zhang, Y. M., & Rock, C. O. (2004). Evaluation of epigallocatechin gallate and related plant polyphenols as inhibitors of the FabG and FabI reductases of bacterial type II fatty-acid synthase. Journal of Biological Chemistry, 279(30), 30994–31001. https://doi.org/10.1074/jbc.M403697200
Zhao, W. H., Hu, Z. Q., Hara, Y., & Shimamura, T. (2002). Inhibition of penicillinase by epigallocatechin gallate resulting in restoration of antibacterial activity of penicillin against penicillinase-producing Staphylococcus aureus. Antimicrobial Agents and Chemotherapy, 46(7), 2266–2268. https://doi.org/10.1128/AAC.46.7.2266-2268.2002
Loading Viewer...
Legal, Permissions, and Contact Information
Author:
Dr. David M Robertson
Wichita, Kansas
Important Note:
While I have expertise in health science, I do not claim expertise in clinical microbiology. This idea and framework emerged from an interdisciplinary analysis regarding another area of interest. That said, the literature review spanned pharmacology, biofilm biology, and redox chemistry. The mechanistic logic appears sound, the compounds are well characterized, and the research gap is verifiable. I merely present this as a theoretical contribution intended to prompt investigation by those with the experimental capacity to test it.
Permissions Notice:
This report is being released directly to the public, not due to a lack of merit but because of the unfortunate constraints typically imposed by academic bureaucracy and disciplinary silos.
This document and its contents, including all associated theories, analyses, and clinical interpretations, are the intellectual property of the author unless otherwise cited. It is provided under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International License unless otherwise noted.
-
You may share this document in its original, unaltered form for non-commercial purposes.
-
You may not alter, republish, or sell any portion of this work without express written permission.
-
Excerpts and citations are allowed with proper attribution to the author and source website: https://www.dmrpublications.com
Collaboration & Inquiries:
To discuss research collaboration, clinical consultations, or publication rights, please contact the author directly at grassfireindustries@gmail.com
Author(s): Dr. David M Robertson
Theoretical Paper | Open Source
Published Online: 2026 Feb – All Rights Reserved. DOI: TBD
APA Citation: Robertson, D. (2026, Feb 20). A Theoretical Framework for EGCG-NAC Combinatorial Strategy Against Biofilm-Protected Bacterial Infections. DMRPublications, https://www.dmrpublications.com/complementary-antagonism-of-biofilm/
Keywords: biofilm, EGCG, N-acetylcysteine, antibiotic adjunct, antimicrobial resistance, combinatorial therapy, disulfide bond disruption, pro-oxidant, thiol conjugate, mixed-species biofilm
Copyright © 2026 – Present. Dr. David M. Robertson, MSL, VL2. All rights are reserved, including those for text and data mining, AI training, and similar technologies.

